U.S. biomedical innovation stands at the forefront of global health advancements, exemplifying the powerful synergy between academia, industry, and government. This innovation ecosystem, which has evolved significantly since World War II, catalyzed breakthroughs such as the mass production of penicillin—saving countless lives during and after the war. Federal funding biomedical research has provided essential resources that fuel this vibrant landscape, promoting public-private research partnerships that continue to drive progress in medical science. As the National Institutes of Health (NIH) plays a critical role in supporting research, historical actions taken during wartime have laid a robust foundation for current and future advancements. Understanding the history of penicillin and the innovative spirit it inspired helps illuminate the ongoing quest for cutting-edge medical solutions in today’s world.
The realm of American health innovation is characterized by transformative breakthroughs and collaborative efforts among various sectors—government, academia, and private industry. This collaborative framework has its origins in urgent wartime needs, particularly during World War II, when the race to develop effective treatments became paramount. The concept of federal investment in scientific research, especially through initiatives like the National Institutes of Health, has been pivotal in establishing a productive research environment that fosters technological advancements. These public-private coalitions have been crucial in leveraging resources for biomedical advancements, which include not just medicines but also health technologies that fundamentally enhance patient care. A deep dive into the historical context of innovations reveals how these partnerships have continuously shaped the landscape of biomedical endeavors in the United States.
The Foundation of U.S. Biomedical Innovation
The roots of U.S. biomedical innovation run deep, tracing back to the pivotal moments of World War II when government-backed research catalyzed a transformation in medical science. During the wartime era, an urgent demand for effective treatments against infectious diseases resulted in the successful mass production of penicillin. This significant breakthrough not only saved countless lives but also laid the groundwork for America’s robust innovation ecosystem in biomedicine. The collaboration between academic institutions and federal entities during this time established a model that would influence medical research and development for decades to come.
The U.S. innovation system emerged from a complex interplay of federal funding, public-private partnerships, and academic research. These elements combined to create a comprehensive model that has since been emulated worldwide. The National Institutes of Health (NIH), although limited at its inception, became a powerhouse of funding and support for biomedical research, allowing universities and private industry to push the boundaries of what was thought possible. The era of penicillin marked the beginning of a golden age in drug development, showcasing how federal investments can lead to breakthroughs in health and medicine.
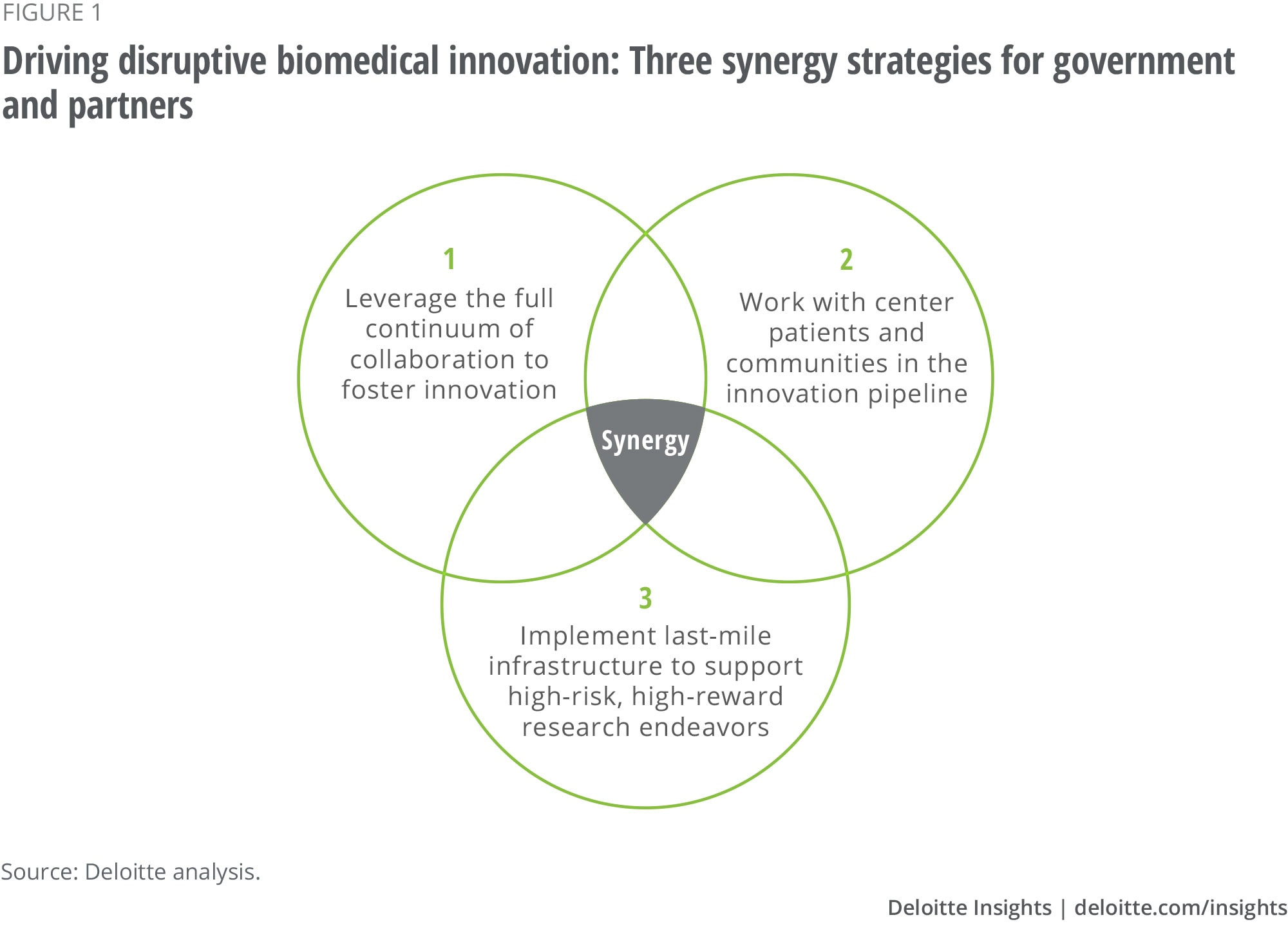
Public-Private Research Partnerships: Key to Innovation
Public-private research partnerships have been essential to the U.S. innovation ecosystem, particularly in the biomedical sector. These collaborations leverage the strengths of both the federal government and the private industry, creating a synergy that drives innovation forward. As highlighted in recent discussions among experts, this partnership model was initially formed to respond to the urgent demands of World War II, culminating in critical developments like penicillin production under the Office of Scientific Research and Development (OSRD). This historical context emphasizes the significance of collaborative efforts in addressing national challenges.
Today, the role of public-private partnerships continues to evolve, with ongoing debates about the allocation of federal funding for biomedical research. The NIH’s support of academic and private endeavors has fostered an environment where innovative solutions can flourish. However, proposed changes to funding policies raise concerns about potential impacts on future research initiatives. Understanding the intricacies of these partnerships is crucial for policymakers and scientists alike as they navigate the balance between public investments and private aspirations in the pursuit of medical advancements.
The Role of Federal Funding in Biomedical Research
Federal funding has been instrumental in transforming the landscape of biomedical research in the United States. The NIH, in particular, plays a crucial role in disbursing these funds, empowering researchers across various institutions to explore new frontiers in medicine. Over the years, federal dollars have led to groundbreaking discoveries and innovations that have drastically improved health outcomes and disease management. The historical backing during World War II, which emphasized quick mobilization of scientific resources, set a precedent for how federal investments can be utilized to spur rapid advancements in health.
However, recent discussions surrounding federal funding cuts, particularly regarding indirect cost reimbursements, highlight the ongoing challenges within this system. These cuts could impact the financial stability of research institutions, ultimately slowing down the pace at which biomedical innovations occur. As the landscape of research funding undergoes scrutiny, it’s vital to recognize the profound implications of federal support and its role in continuing to sustain the U.S. biomedical innovation ecosystem.
Historical Context: The Legacy of Penicillin
The story of penicillin is a remarkable chapter in the history of medicine that underscores the interplay between innovation and the pressing needs of society. Discovered by Alexander Fleming in the 1920s, it wasn’t until World War II that efforts to mass-produce this vital antibiotic accelerated. The urgency of the war highlighted the necessity for effective treatment against bacterial infections, which often debilitated soldiers more than injuries from combat. The wartime collaboration between the military and scientific community not only led to the successful production of penicillin but also demonstrated the potential of scientific innovation when supported by focused investment and cooperation.
This legacy of collaboration continues to influence biomedical research today. The advances made during this period set the stage for extensive pharmaceutical research and development, resulting in a vast array of antibiotics and other medications that have fundamentally changed medical practice. Understanding the history of penicillin serves as a reminder of the profound impact that strategic partnerships and federal funding can have on public health, paving the way for future innovations that address similar challenges.
World War II: A Catalyst for Biomedical Progress
World War II was not just a time of conflict; it was also a crucial period that sparked substantial advancements in biomedical research and technology. The war prompted a re-evaluation of existing medical practices, showcasing the need for innovative solutions to combat infectious diseases that threatened military effectiveness. The establishment of agencies like the OSRD was pivotal in coordinating wartime research efforts, ultimately leading to breakthroughs that would later benefit civilian populations as well. This collective urgency to combat diseases laid the groundwork for an unprecedented era of medical discovery.
The technological advancements borne out of wartime research significantly influenced post-war biomedical initiatives. The wartime experience fostered an environment where scientists and researchers were encouraged to push the boundaries of traditional medical approaches, leading to the establishment of modern methodologies and practices in drug discovery and development. The foundation laid during this period inevitably evolved into the sophisticated U.S. biomedical innovation system we witness today, characterized by collaborative efforts, substantial federal funding, and a thriving public-private research landscape.
Future Directions for U.S. Biomedical Innovation
As we look to the future of U.S. biomedical innovation, it is imperative to maintain the delicate balance between public and private interests. The existing ecosystem has produced remarkable advancements in medicine and technology, yet emerging challenges—such as rapid technological changes and shifts in funding policies—demand adaptive strategies. The continued success of the U.S. innovation system hinges on the synergies formed through public-private partnerships that drive research forward while effectively addressing the complex healthcare needs of society.
Moreover, advocating for sustainable federal funding models is essential for fostering ongoing innovation in biomedicine. As discussions around reimbursement for indirect costs and research funding continue, it is vital to ensure that the momentum of scientific inquiry remains unimpeded. Future initiatives should strive to uphold the collaborative spirit established during World War II, fostering environments where all stakeholders in the biomedical landscape can work together to harness new technologies and improve patient outcomes.
The Importance of Scientific Training in Biomedical Research
Scientific training plays a critical role in sustaining the momentum of innovation within the U.S. biomedical system. During World War II, the engagement of thousands of young researchers helped catalyze breakthroughs that would reshape medical science. The influx of new talent not only contributed to immediate wartime needs but also established a pipeline of skilled scientists equipped to tackle future challenges in health and medicine. The importance of investing in the next generation of biomedical researchers cannot be overstated, as they carry the legacy of innovation forward.
In contemporary settings, fostering rigorous training programs within research institutions is essential for maintaining the high standards of biomedical research. Both public and private sectors must prioritize the development of young scientists, ensuring that they receive the necessary guidance, resources, and support to flourish in their fields. By enhancing scientific training, we can ensure the U.S. remains at the forefront of biomedical innovation, continuously producing impactful discoveries that improve global health outcomes.
Navigating Challenges in Biomedical Research Funding
The landscape of funding for biomedical research is fraught with complexities and challenges that require careful navigation. As debates on federal funding models intensify, the implications for public-private partnerships become increasingly significant. Researchers and institutions are seeking clarity on how changes in funding policy could affect their ability to conduct pivotal studies and deliver innovative solutions. The tension between budget restraints and the need for robust research funding necessitates a critical examination of existing structures to ensure a sustainable future for biomedical inquiry.
Addressing these challenges will require collaboration among policymakers, academic leaders, and industry stakeholders. By re-evaluating funding mechanisms and recognizing the vital role of public investment in scientific research, we can foster an environment where biomedical innovation thrives. Ensuring a stable funding stream not only supports ongoing research efforts but also enhances the potential for groundbreaking discoveries that can have far-reaching implications for healthcare globally.
Ensuring Sustainable Biomedical Innovation Policy
Sustaining the momentum of biomedical innovation in the U.S. hinges on proactive policy frameworks that embrace collaboration between government, academia, and the pharmaceutical industry. The historical partnership formed during World War II serves as a blueprint for modern-day strategies aimed at fostering innovation. Policymakers must advocate for consistent funding models that empower scientific research while balancing budgetary constraints. This commitment to clear and supportive policy can provide the foundation necessary for ongoing advancements in health technology and medical treatments.
Furthermore, engaging a diverse array of stakeholders in the policy-making process can lead to more inclusive and effective solutions that address the multifaceted challenges of biomedical research. Fostering dialogue among researchers, clinicians, and industry leaders will yield insights that shape a proactive approach to maintaining America’s position as a global leader in biomedical innovation. By prioritizing sustainable policies that recognize the critical importance of investment in research and development, we safeguard the future of medical breakthroughs that benefit society.
Frequently Asked Questions
How has World War II influenced U.S. biomedical innovation?
World War II had a profound impact on U.S. biomedical innovation by fostering a public-private research partnership that prioritized rapid technological advancement. The urgent need for solutions to health crises, such as infectious diseases that affected military personnel, led to significant investments in biomedical research. This partnership laid the groundwork for future advancements, including the mass production of penicillin, ultimately shaping the modern U.S. biomedical innovation ecosystem.
What role does federal funding play in U.S. biomedical innovation?
Federal funding is crucial for U.S. biomedical innovation as it supports research projects in universities and private industries. This funding, primarily through agencies like the National Institutes of Health (NIH), catalyzes groundbreaking discoveries in medicine and technology. The historical significance of federal funding can be traced back to World War II, where it initiated a robust collaboration between government and academia, leading to advancements such as penicillin.
What are the key components of the public-private research partnership in U.S. biomedical innovation?
The public-private research partnership in U.S. biomedical innovation involves collaboration between federal agencies, private industries, and academic institutions. This ecosystem emerged during World War II to address urgent health needs and has evolved to include shared funding, expertise, and resources, fostering groundbreaking research and development in biomedical sciences.
How did the history of penicillin impact modern U.S. biomedical innovation?
The history of penicillin is a cornerstone in U.S. biomedical innovation, highlighting the significance of public-private partnerships that emerged during World War II. Efforts to mass-produce penicillin not only enhanced military health but also paved the way for the antibiotic era in civilian health, demonstrating the powerful outcomes that result from collaborative research efforts.
Why is NIH research funding considered vital for biomedical innovation in the U.S.?
NIH research funding is vital for U.S. biomedical innovation as it provides essential financial support for academic and clinical research. This funding enables the discovery of new treatments and therapies, directly impacting public health. Historical parallels can be drawn to World War II, where NIH-like initiatives backed the scientific efforts necessary to produce breakthrough medical advancements.
What challenges does the U.S. biomedical innovation ecosystem face today?
Today, the U.S. biomedical innovation ecosystem faces challenges such as potential funding cuts from federal sources, like the NIH, and rising questions about the sustainability of public-private partnerships. These challenges could hinder the progress and breakthroughs that the robust system nurtured since World War II has produced, emphasizing the need to protect and adapt this successful collaboration.
How did World War II innovation shape the future of biomedical sciences in the U.S.?
World War II innovation significantly shaped the future of biomedical sciences by establishing a model of federal investment in research and the collaboration between government and private sectors. The focus on urgent health solutions during the war, particularly regarding penicillin, not only saved lives but also formed the framework for subsequent advancements in medical research and technology.
What is the importance of public R&D funding for training the next generation of biomedical scientists?
Public R&D funding is crucial for training the next generation of biomedical scientists as it provides resources for educational opportunities, research positions, and hands-on experiences in laboratories. The legacy of World War II, with its emphasis on engaging young scientists, continues to influence how federal funding supports both research and the cultivation of future leaders in biomedical innovation.
In what ways can the U.S. innovation system be improved to enhance biomedical research?
To enhance biomedical research, the U.S. innovation system could be improved by increasing collaboration across sectors, streamlining funding processes, and ensuring sustained investment in emerging research areas. Learning from the successes of the World War II era public-private partnerships, reforming policies to support innovation while preserving funding mechanisms like the NIH could further drive breakthroughs in health.
| Key Points | Details |
|---|---|
| Historical Context | The U.S. biomedical innovation system originated during World War II, driven by the urgent need for new medical technologies and treatments. |
| Government Role | Federal funding has been fundamental in supporting academic research, which in turn fosters private sector development. |
| Penicillin Production | The mass production of penicillin during World War II marked a significant breakthrough in healthcare and saved countless lives, showcasing the effectiveness of public-private partnerships. |
| Public-Private Partnerships | The collaboration between government, academia, and industry has been vital for technological advancements in biomedicine over the last 80 years. |
| Impact on Modern Research | The infrastructure established during WWII laid the foundation for present-day biomedical research and drug development. |
| Current Landscape | Despite recent challenges, the U.S. biomedical innovation system remains pivotal for health and economic growth, making it a global leader. |
Summary
U.S. biomedical innovation has emerged as a crucial driver of health advancements and economic stability, rooted in its historical partnership between government agencies and academic institutions. This collaboration, which traces back to World War II, has fostered groundbreaking research, especially in developing life-saving drugs like penicillin. The effectiveness of this public-private synergy has attracted international admiration, underscoring the importance of maintaining strong federal support to ensure continued success in biomedical research. As the global landscape evolves, preserving this collaborative framework will be essential in addressing ongoing health challenges and sustaining U.S. leadership in biomedical innovation.